 
Among them hydrogen bonding interactions are crucial because of their energy parameters and directionality ( Gamekkanda et al., 2018). Generally, cocrystals rely on various intermolecular interactions like hydrogen bonding, π-π stacking and van der Waals forces. Cocrystals present an opportunity to enhance the physicochemical properties like solubility, stability, bioavailability, dissolution rates, compressibility and hygroscopicity without altering chemical structure of active pharmaceutical ingredients (API) ( Corner et al., 2018 Alhalaweh et al., 2012 Jones et al., 2006 Shimpi et al., 2018 Bolla and Nangia, 2016). Pharmaceutical cocrystals ( Aakeröy and Sinha, 2018 Duggirala et al., 2016) have received interest in recent years from a variety of disciplines including chemical, material and pharmaceutical sciences. The results show that the physicochemical properties of CAF-CA cocrystal are enhanced in comparison to CAF (API). The molar refractivity value of cocrystal lies within the set range by Lipinski and hence it may be used as orally active form. plotting molecular electrostatic potential (MESP) map. 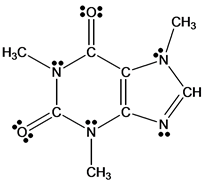
The electrophilic and nucleophilic reactivities of atomic sites involved in intermolecular hydrogen bond interactions in cocrystal have been demonstrated by mapping electron density isosurfaces over electrostatic potential i.e. The HOMO-LUMO energy gap value shows that the CAF-CA cocrystal is more reactive, less stable and softer than CAF active pharmaceutical ingredients. The natural bond orbital analysis of the second-order theory of the Fock matrix highlighted the presence of strong interactions (N∙∙∙H and O∙∙∙H) in cocrystal. The quantum theory of atoms in molecules (QTAIM) analysis revealed (O88-H89∙∙∙O41) as strongest intermolecular hydrogen bonding interaction with interaction energy −12.4247 kcal mol −1 in CAF-CA cocrystal. The spectroscopic results confirmed that (O33-H34) mode forms an intramolecular (C25 = O28∙∙∙H34-O33), while (O26-H27) (O39-H40) and (O43-H44) groups form intermolecular hydrogen bonding (O26-H27∙∙∙N24-C22, O39-H40∙∙∙O52 = C51 and O43-H44∙∙∙O86 = C83) in cocrystal due to red shifting and increment in bond length. 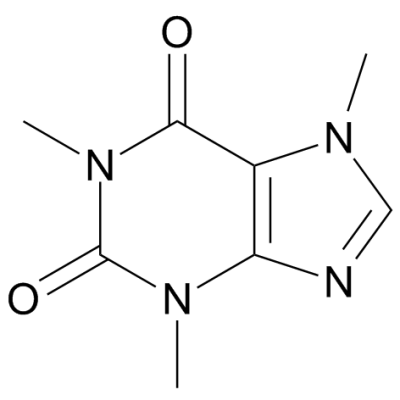
There was a good correlation among the experimental and theoretical results of dimer of cocrystal, as this model is capable of covering all nearest possible interactions present in the crystal structure of cocrystal. The vibrational spectroscopic (FT-IR and FT-Raman) signatures and quantum chemical approach have been used as a strategy to get insights into structural and spectral features of CAF-CA cocrystal. Also, differential scanning calorimetry (DSC) confirmed the existence of CAF-CA cocrystal. Powder X-ray diffraction (PXRD) analysis was done to characterize CAF-CA cocrystal. The cocrystal was prepared by slurry crystallization. The pharmaceutical cocrystal of caffeine-citric acid (CAF-CA, Form II) has been studied to explore the presence of hydrogen bonding interactions and structure-reactivity-property relationship between the two constituents CAF and Citric acid. 4Chemistry of Interfaces, Luleå University of Technology, Luleå, Sweden.3Department of Materials and Environmental Chemistry, Stockholm University, Stockholm, Sweden.2Department of Physics, Isabella Thoburn (I.T.) College, Lucknow, India.1Department of Physics, University of Lucknow, Lucknow, India.Priya Verma 1 Anubha Srivastava 1 Karnica Srivastava 2 Poonam Tandon 1* Manishkumar R. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
